
NEW DELHI — Two government-appointed committees have flagged concerns about the reliability and capability of 10,000 low-cost ventilators, ordered by the Narendra Modi government as part of India’s response to the Covid-19 pandemic, according to documents reviewed by HuffPost India.
In a clinical evaluation report dated June 1, 2020, a committee of doctors said that the Modi government could buy these ventilators, made by Indian startup AgVa Healthcare, but added that AgVa’s Covid-model ventilators “should not be considered as a replacement for high-end ventilators in tertiary care ICUs”.
The experts also said, “There should be a provision for backup ventilator in the facility where these ventilators are used.”
The June 1 report was the second time AgVa ventilators were being evaluated by the government. Two weeks previously, on May 16, another clinical evaluation committee had concluded that AgVa’s devices needed “further technical validation” before they could be approved.
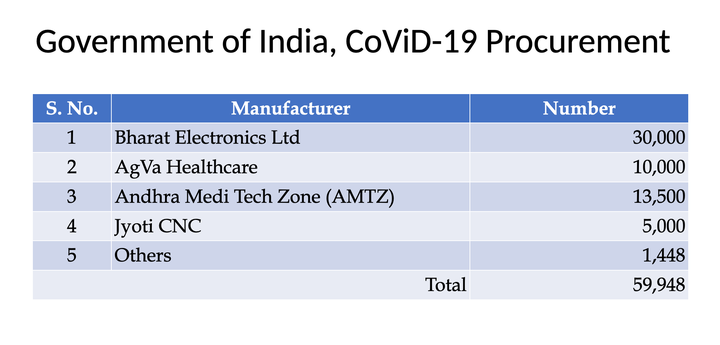
The order for 10,000 AgVa ventilators was placed on March 27 as part of a union government plan to procure 50,000 ventilators for hospitals to cope with an anticipated surge of Covid patients. The ventilators were bought under the controversial PMCARES fund.
Given the global shortage of ventilators, Indian companies were invited to bid on the condition that once an order was placed, the government would accept the devices only after they were approved by an expert panel.
In AgVa’s case, the committee’s qualified approval raises the question of why the Modi government is buying these ventilators at all. The committee’s caveats — that the ventilators are not a replacement for ICU-grade ventilators, and should be used in facilities that have backup ventilators — are specific to AgVa devices.
Defence PSU Bharat Electronics Limited is producing 30,000 ventilators for the government in technical collaboration with Skanray Technologies, another Indian company. Skanray Technologies said their ventilators had been evaluated and approved by the government without any preconditions.
“No such guidelines have been issued to us,” Skanray’s managing director Vishwaprasad Alva said in an interview.
HuffPost India reviewed AgVa’s internal company chats, interviewed two former AgVa employees and spoke to doctors familiar with AgVa products. Our findings mirror the concerns raised by the government experts who evaluated AgVa’s ventilators, and uncover previously unreported performance issues with AgVa devices.
AgVa has contested HuffPost India’s interpretations of the committee reports. AgVa cofounder Diwakar Vaish insisted that his ventilator is suitable for tertiary ICUs, despite the government’s clinical evaluation committee concluding otherwise.
Vaish said the company had fixed all issues raised by the clinical evaluation committees, and that the company had built all 10,000 ventilators and was awaiting dispatch orders.
“AgVa Ventilators satisfy point by point all essential specifications for a high-end ventilator in tertiary care ICU as drafted by the expert committee and there is no controversy on this point,” Vaish said over email.

“We would also like to point out that we were competing for the same specifications with other extremely expensive ventilators costing upwards of 20 lakhs a piece. As our ventilator is disrupting the ventilator eco-system with aggressive pricing and feature set, it has not gone down well with the Doctor-Vendor nexus which wants to manage status quo of having extremely expensive ventilator care in ICUs.”
HuffPost India also sent detailed questionnaires to HLL Lifecare Ltd, the public sector unit handling the ventilator tender for the Indian government, and the Department of Pharmaceuticals which is overseeing the process, but did not hear back from them.
Independent experts said the expert committee reports — that said AgVa ventilators were not replacements for ventilators in tertiary care ICUs, and should not be used without a backup — called for more discussion.
“The reason you are buying these is because you don’t have ventilators. Even if you say 5 backups for ten machines, where will you get those backup ventilators?” asked Dr. Anant Bhan, a physician and former president of the International Association of Bioethics. “There is already a shortage. So if you are procuring this for the public sector, one needs to ask what is the function these ventilators are performing.”
“All of this data should be transparently available. Quality measures can’t be bypassed during a pandemic,” said Dr. Bhan. “Evaluation reports should be in the public domain.”
The ‘Covid model’
When the Indian government frantically sought to buy tens of thousands of ventilators in March this year, AgVa Healthcare became a poster child for the Modi administration’s decision to source ventilators locally by pairing Indian start-ups with manufacturing powerhouses. AgVa was selected by auto giant Maruti Suzuki from a list of approved ventilator manufacturers shortlisted by the Indian government.
The tie-up between AgVa and Maruti Suzuki was accompanied by a flurry of publicity by the companies involved, and also by the Modi government, which was eager to show progress in the fight against the novel coronavirus. NITI Aayog CEO Amitabh Kant, for instance, wrote at least three articles in which he name-checked AgVa ventilators.
Collaborating with Maruti Suzuki and winning a big government order conferred an aura of legitimacy on AgVa — a startup striving to break into a ventilator market dominated by established global firms. Charitable foundations eager to contribute to India’s coronavirus effort bought and donated AgVa ventilators in large numbers.
The collaboration did not involve any financial arrangements, AgVa and Maruti Suzuki confirmed to HuffPost India; the auto company would bring its assembly line expertise to help AgVa massively scale up production to a peak capacity of 600 ventilators a day.
The order for 10,000 ventilators is AgVa’s largest order ever. For context, Vaish told HuffPost India that AgVa had installed about 1,500 ventilators in the 2 years preceding this order.
“Maruti Suzuki decided to help AgVa because it had a product approved by the buyer and had an order for 10,000 ventilators from HLL. Further it was situated in the NCR and so it was easier for us to assist manufacturing,” a Maruti Suzuki spokesperson told HuffPost India over email. “This was done as Govt Of India asked Maruti Suzuki to help in increasing production of ventilators.”
The spokesperson said the carmaker was only helping AgVa streamline its production lines. “Maruti Suzuki is not responsible for technology, performance or commercial issues.”
AgVa’s ventilators were co-invented by Dr. Deepak Agrawal, a neurosurgeon at the All India Institute of Medical Sciences, and Vaish, a robotics engineer, with the aim of disrupting the medical device market. High-end ICU-grade ventilators routinely cost tens of thousands of dollars. AgVa’s ventilators, which the company claims are as good as their more expensive competitors, cost upto Rs 2.5 lakh or about $3,300.
AgVa’s early models were meant for neurology patients, who had mostly healthy lungs but still needed ventilator support. Over time, the company developed more sophisticated models that the company claimed could be used at home, in emergency ambulances, and in critical care facilities, or ICUs.
When the government put out a tender in March for ventilators to treat Covid patients, AgVa said they quickly developed a Covid model.
“We added a few features to the high-end ventilator,” Dr. Deepak Agrawal, the co-inventor of the ventilator, told HuffPost India. “This added to the price and we called it the Covid model.”
Agrawal said the company also improved the software, adding that software updates were an ongoing process. “That is the heart of the ventilator,” he said. “So we improve the algorithms and the AI powering it.”
A well-placed source familiar with the deliberations of both committees said that it was not possible to develop a reliable high-specification ventilator in such little time.
“There is a big difference between a home ventilator or an emergency ventilator meant for patients whose lungs are mostly functional, and a ventilator for Covid — which is a disease that destroys the lungs,” said the source, who has years of experience with ventilators and is familiar with the product. The source sought anonymity as he was not authorised to speak with the press.
Cleared, but with caveats
AgVa has consistently claimed that their Covid model meets all specifications listed in the government tender, but in clinical trials, government committees found problems.
The first evaluation committee, which submitted its report on May 16 based on trials conducted on patients at the Ram Manohar Lohia hospital in New Delhi, said that AgVa’s ventilator wasn’t maintaining respiratory parameters such as the volume of gas pumped into a patient’s lungs, the rate at which gas is pumped into the lungs, and a technical parameter called PEEP, or Positive End-Expiratory Pressure.
The committee concluded: “This requires further technical validation. This ventilator should be put to use in multicentre multiple diseases patients with variety of end users to prove its versatility, ruggedness and patient safety. Then this ventilator will be suitable as emergency ventilator and settings where MGPS is not available.”
MGPS, or a Medical Gas Pipeline System, refers to a centralised gas supply installed in well-equipped hospitals. Emergency ventilators in ambulances, and in smaller district hospitals in India, use oxygen stored in cylinders.
Ten days later, on May 27, a re-evaluation committee was appointed to re-examine AgVa’s ventilator. This committee said that AgVa had fixed the problem of PEEP variation, and accepted AgVa’s explanations for many of the issues raised by the previous committee.
The re-evaluation committee cleared the ventilators on June 1. A note at the end of their observations said the device was being cleared as “considering the covid situation it is necessary to have ventilators available in adequate numbers throughout the country.”
AgVa’s ventilators, the note said, “should be purchased in phased manner and not all together. Feedback from end users may be taken before further procurement.”
“It should not be considered as a replacement for high-end ventilators in tertiary care ICUs,” the note added.
The note was followed by a set of guidelines. Guideline No.1 said, “There should be a provision for backup ventilator in the facility where these ventilators are used.”
“On the face of it, this is a strange thing,” said Dr. Bhan, the bioethicist, explaining that ventilators are primarily used in tertiary care facilities. “Maybe you can use it in secondary care if you have trained staff, but mostly it is for tertiary care.”
AgVa responds
AgVa disputed HuffPost India’s characterisation of the expert committee reports. The problems observed by the first committee, Vaish said, were because the committee —staffed with experienced doctors — had not used the correct settings for the ventilator.
The variation in PEEP, for instance, Vaish said, had been fixed in the following way:
“We have developed a new algorithm that warns the user through suggestive alarms if any incorrect settings in the ventilator is done. Further we have developed an algorithm that despite wrong settings can still maintain a stable PEEP.”
“The ventilator was working just fine,” Vaish said over email.
Vaish also contested the government committee’s assessment that AgVa ventilators were not good enough for tertiary-care ICUs.
“Our core speciality is cost-effective ventilators in ALL categories,” he said. “We would like to reiterate that the doctor-vendor nexus present in our country is not allowing home grown quality products to survive.”
The committee’s requirement that AgVa ventilators be used with backups was explained in the following manner.
“This does not imply having a ventilator of a different make as a backup,” Vaish said. “Having two AgVa ventilators will also satisfy the requirements of having a backup ventilator and is a normal requirement as back up ventilation facilities should always be available.”
Long-pending issues
A former AgVa employee told HuffPost India that AgVa ventilators had issues that pre-date the Covid model.
On February 25, a month before AgVa bagged its big government order for 10,000 ventilators, doctors at Prayag Hospital in Noida told AgVa sales representatives that their ventilator was not maintaining key ventilator parameters like tidal volume, and unnecessary alarms in the night caused them to remove the ventilator according to WhatsApp chats reviewed by HuffPost India.

Dr. Zameer Farooq, an anesthesiologist and critical care surgeon at Prayag Hospital, confirmed to HuffPost India that AgVa had installed a ventilator at their hospital for use on a trial basis.
The AgVa ventilator was connected to a patient in their ICU, but the hospital disconnected it when they found the device could not maintain the Tidal Volume — the amount of air delivered by the ventilator in each breath — and kept sounding alarms. The patient was taken off the AgVa ventilator in the night and put onto the hospital’s regular ventilator, and the situation was controlled, Dr. Farooq said.
The expert committee appointed by the government had made a similar observation about AgVa’s Covid model in their May 16 2020 report. “Tidal volume varied from 400 to 525 ml when it was fixed at 500 ml,” the report said.
“We didn’t have any positive feedback to share,” Dr. Farooq from Prayag Hospital said, describing his experience with AgVa. “So we returned the ventilator the next day.”
Vaish agreed that the AgVa ventilator at Prayag hospital did not deliver the right tidal volume, but blamed the hospital for negligence.
“Your reporting is absolutely correct on the point that Tidal volume was not being delivered,” Vaish said, but said this was due to a leak in the “cuff” which is fitted in a patient’s throat to ensure the ventilator works properly.
“Whenever a ventilator is put on a patient there should always be an anaesthetist/ intensivist to control the ventilator. However Prayag hospital had only visiting anaesthetist/intensivist who were not present at night,” Vaish said. “Had our ventilator not raised an alarm the patient could have died without the much needed intervention. So in a way our ventilator saved a life.”
Dr. Farooq was indignant in his response. He said there was no cuff leakage, as Vaish had claimed, and their anaesthetist was on duty at the time.
“How can they make this statement that there is no intensivist?” Dr. Farooq said. “We have a 24X7 anaesthetist in our ICUs. You can come and check our registers. This is absolutely false.”
Prayag Hospital’s experience, the former employee said, was not unusual.
“After every demo or doctor conference, we used to come and brief the company of the problems and issues raised,” the former employee said, adding that clients often complained about falling tidal volume.
A month prior to the incident at Prayag Hospital, five of eight AgVa ventilators installed at the Subharti hospital in Meerut stopped working, according to internal company chats reviewed by HuffPost India.
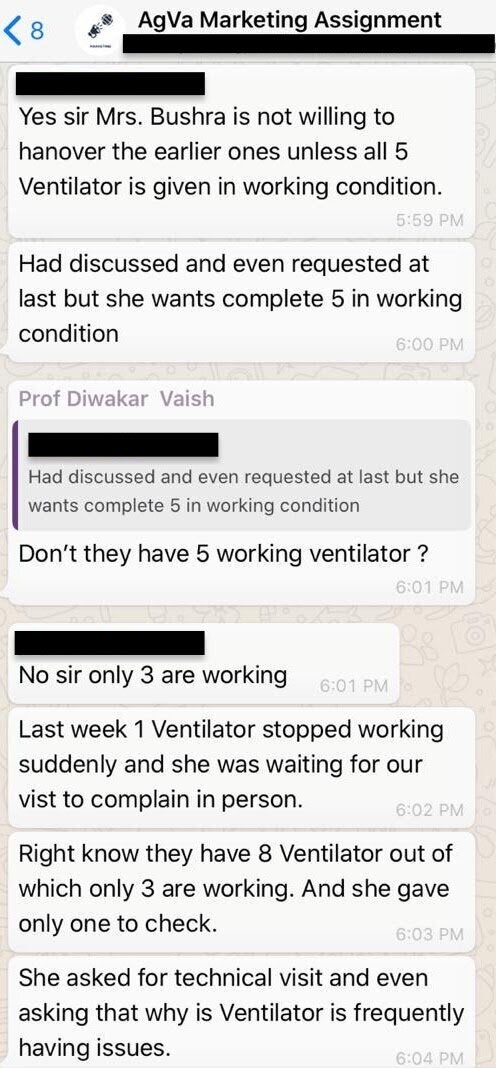
The hospital administrator was frustrated and asked the employees why the ventilator was “frequently having issues”, according to the chat logs, dated January 29, 2020.
In his email, Vaish agreed that the ventilators had stopped working, but blamed the hospital once more.
“The ventilators malfunctioned due to supply of humidified oxygen. Any ventilator in the world has to be supplied with dry oxygen and not humidified,” Vaish said. “Despite of this we provided replacements out of courtesy.”
HuffPost India shared Vaish’s explanation with Subharti Hospital, but did not hear back.
On May 18, the Press Trust of India quoted Ahmedabad Civil Hospital Medical Superintendent JV Modi as saying that the hospital was not getting “desired results” when AgVa ventilators were placed on Covid patients.
On June 19, the Mumbai Mirror reported that doctors in two prestigious Mumbai Hospitals, JJ Hospital and St George Hospital, had refused to use 81 AgVa ventilators as they were not suited to treat Covid patients.
Dr TP Lahane, who heads the Directorate of Medical Education, told the Mirror that AgVa’s ventilators could, at best, be used while transferring patients — the role they were originally designed for.
When HuffPost India reached out to Dr. Lahane, he declined to make any further comments on AgVa.
AgVa, as always, insisted their devices were working perfectly.
Aman Sethi is the Editor-in-Chief of HuffPost India. You can write to him at aman.sethi[at]huffpost.in